Products
Showing 19–36 of 74 items
Cupric Sulphate
CAS NO: 7758-99-8
MOLECULAR FORMULA : CuH10O9S
MOLECULAR WEIGHT : 249.69
SPECIALITY FINE CHEMICALS (AR / LR / ACR /GR / IP / BP / USP / FSSAI )Dibasic Potassium Phosphate
CAS NO:7758-11-4
MOLECULAR FORMULA : K2HPO4
MOLECULAR WEIGHT : 174.18
SPECIFICATION BP USP Description White or almost white powder or colorless crystals, very hygroscopic. White or almost white powder or colorless crystals, very hygroscopic. Solubility Very soluble in water, very slightly soluble in ethanol (96 per cent). – Appearance of solution Solution is clear & Colorless – Identification (A) ) Reaction
(B) Reaction
(C) Reaction
A. Solution S (see Tests) is slightly alkaline B. Solution S gives reaction of phosphate
C. Solution S gives reaction of potassium
A. Solution S gives reaction of phosphate B. Solution S gives reaction of potassium
Reducing substances The solution remains faintly pink. – Heavy Metals – NMT 0.001 % Limit of Fluoride – NMT 0.001 % pH – 8.5-9.6 Monopotassium phosphate This ratio is not greater than 0.025. – Chlorides NMT200 ppm – Sulphates NMT 0.1 % – Arsenic NMT 2 ppm – Sodium NMT 0.1 % A solution tested on a platinum wire imparts no pronounced yellow color to a nonluminous flame. Limit of Monobasic OR tribasic salt – NMT 0.001 % Iron NMT10 ppm NMT 30 ppm Residual solvents – Meets the requirement Loss on Drying(at 125-130˚C) NMT 2.0 % NMT 1.0 % Assay (dried substance). 98.0 % to 101.0 % 98.0 % to 100.5 % Direct Blues 86
1%4%DIRECT TURQ. BLUE STD (Direct Blue 86)DIRECT TURQ. BLUE 135%. (Direct Blue 86)General Properties Dyeing
PropertiesExhaustionFairLevelingGoodFastness
PropertiesAcid3Alkaline3Hot Pressing3-4Day Light 1/16-7WashingAlteration2Stain3WaterAlteration2Stain3-4DischargeabilityNetural
Fairy Good
Alkaline-
FairFixation Temp. Exhaust Dyeing90°CSolubility30-40Salt and Alkali RequirementDepth of shade % (o.w.f.)Salt (g/l)Soda (g/l)Up to 0.5100.50.5 – 1.015-2012.0 – 4.020-251.5Above 4.025-302.0Exhaust Dyeing

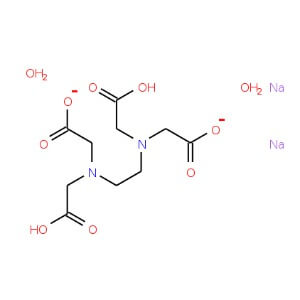
Disodium Edetate
CAS NO: 6381-92-6
MOLECULAR FORMULA :- C10H14N2Na2O82H2O
MOLECULAR WEIGHT :- 372.24 g/ mol
SPECIFICATION IP BP USP Description A white ,crystalline powder, Odourless White or almost white, crystalline powder. White or almost white, crystalline powder. Solubility – Soluble in water, practically insoluble in ethanol (96 %) – Identification A. IR
B. Reaction
C. Reaction
D. Reaction
A. Must comply with the disodium edetate RS
B. No precipitate is produced.
C. No precipitate is produced.
D. It gives reaction of sodium salt.
A. Must comply with the disodium edetate RS
B. No precipitate is produced.
C. No precipitate is produced.
D. It gives reaction of sodium salt.
A. Must comply with the disodium edetate RS
B. The red color is discharge, leaving a yellowish solution
C. It gives reaction of sodium salt.
Appearance of solution 5 % w/v solution in carbon dioxide free water is Clear & colorless Clear & colorless – pH 4.0-5.5 ( 5 % w/v solution ) 4.0-5.5 ( 5 % w/v solution ) 4.0-6.0 Loss on Drying – – 8.7 %-11.4 % Calcium – – No precipitate is formed. Impurity A Limit of nitrilotriacetic acid(USP)
NMT 0.1 % NMT 0.1 % NMT 0.1 % Heavy metals NMT 20 ppm – NMT 50 ppm Iron NMT 80 ppm NMT 80 ppm – Assay 98.5 %-101.0 % 98.5 %-101.0 % 99.0 %-101.0 %(on Dried basis) Dried Ferrous Sulphate
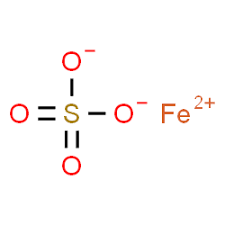
CAS NO: 7720-78-7
MOLECULAR FORMULA :- FeSO4
MOLECULAR WEIGHT :- 151.91 g/mol
SPECIFICATION IP BP USP Description Grayish white to buff colored powder. Grayish white powder. Grayish white powder. Solubility – Slowly but freely soluble in water. Very soluble in boiling water, practically insoluble in ethanol (96%) – pH – 3.0-4.0 – Identification (A) Reaction
(B) Reaction
(C) Assay
A. Reaction of sulfates B. Reaction of Ferrous Salt
A. Reaction of sulfates B. Reaction of Irons
C. It complies with limit of assay.
A. Reaction of sulfates B. Reaction of Ferrous Salt
Chloride – NMT 300 ppm – Insoluble Substances – – NMT 0.05 % Basic sulphate Producing solution that is not more than faintly turbid. – – Arsenic NMT 3 ppm – NMT 3 ppm Lead NMT 50 ppm – NMT 10 ppm Mercury – – It meets the requirements of the test for mercury Chromium – NMT 100 ppm – Copper NMT 50 ppm NMT 50 ppm – Ferric ions – NMT 0.5 % – Manganese NMT 0.1 % NMT 0.1 % – Nickel – NMT 100 ppm – Zinc NMT 500 ppm NMT 100 ppm – Organic volatile impurities – – Meets the requirements. Residual Solvents – – Meets the requirements. Assay 86.0 % – 90.0 % 86.0 % – 90.0 % 86.0 % – 89.0 % Ferric Ammonium Citrate
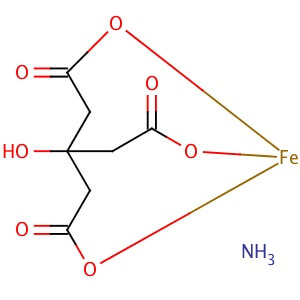
CAS NO: 1185-57-5
MOLECULAR FORMULA : C6H8O7•xFe(III)•yNH3
MOLECULAR WEIGHT :
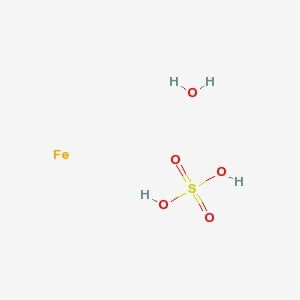
SPECIALITY FINE CHEMICALS (AR / LR / ACR /GR / IP / BP / USP / FSSAI )Ferrous Sulfate Heptahydrate
CAS NO: 7782-63-0
MOLECULAR FORMULA :- FeSO4.7H2O
MOLECULAR WEIGHT :- 278.02 g/mol
SPECIFICATION IP BP USP Description Bluish green crystals or a light green, crystalline powder, odourless.Efforescent in air. On exposure To moist air, the crystals rapidly oxidize and become brown. Bluish green crystals or a light green, crystalline powder,efforescent in air. Bluish green crystals or a light green, crystalline powder, odourless.Efforescent in air. Solubility – Freely soluble in water, very soluble in boiling water, practically insoluble in ethanol (96 %) Ferrous sulfate heptahydrate is oxidized in moist air, becoming brown. – pH 3.0-4.0 ( 5 % w/v Solution) 3.0-4.0 – Appearance of Solution The solution is not more opalescent than opalescent Standard. – – Identification (A) Reaction
(B) Reaction
(C) Assay
A. It gives reaction of sulfates B. It gives reaction of Ferrous Salt
A. It gives reaction of sulfates B. It gives reaction of Irons
C. It complies with limit of assay.
A. It gives reaction of sulfates B. It gives reaction of Ferrous Salt
Chloride NMT 250 ppm NMT 200 ppm – Basic sulphate Producing solution that is not more than turbid. – – Arsenic NMT 2 ppm – NMT 3 ppm Lead NMT 50 ppm – NMT 10 ppm Mercury – – It meets the requirements of the test for mercury Chromium – NMT 50 ppm – Copper NMT 50 ppm NMT 50 ppm – Ferric ions NMT 0.5 % NMT 0.3 % – Manganese NMT 0.1 % NMT 0.1 % – Nickel – NMT 50 ppm – Zinc NMT 500 ppm NMT 50 ppm – Organic volatile impurities – – Meets the requirements. Residual Solvents – – Meets the requirements. Assay 98.0 % – 105.0 % 98.0 % – 105.0 % 99.5% -104.5 % Magnesium Chloride Hexahydrate
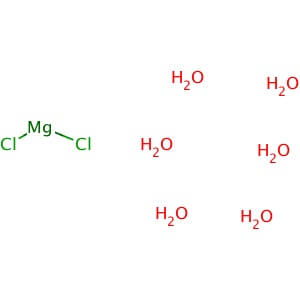
CAS NO: 7791-18-6
MOLECULAR FORMULA: MgCl2•6H2O
MOLECULAR WEIGHT: 203.30
Application: Magnesium Chloride, Hexahydrate is a source of magnesium ion and a co-foactor for many enzymes Purity: >99% Molecular Weight: 203.3 Molecular Formula: MgCl2•6H2O Description: Magnesium Chloride, Hexahydrate is widely used as a source of magnesium ion in chemistry and molecular biology applications. In biological systems, magnesium is a co-factor for many enzymes including deoxyribonuclease (DNase) and various restriction enzymes. Also plays a role in cell membrane integrity, muscle cell physiology, cardiovascular and muscular activity, and nucleic acid structure. Magnesium chloride solution is a favorable choice as an elution buffer for antibody affinity column purifications; it is much milder on most antigens than acid elution, allowing reuse of the antigen column. Also an essential cofactor for the DNA polymerase in polymerase chain reaction (PCR) amplification. Physical State : Solid Solubility : Soluble in water (20 mg/ml), and alcohol. Storage : Store at room temperature Melting Point : 116-118° C (dec.) Density : 1.57 g/cm3 at 20° C Magnesium Sulfate Heptahydrate
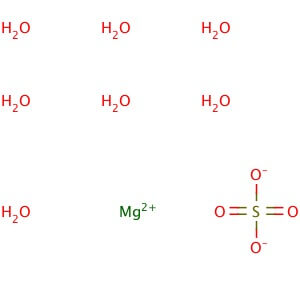
CAS NO: 10034-99-8
MOLECULAR FORMULA :- MgSO4.7H20
MOLECULAR WEIGHT :- 246.50 g/mol
SPECIFICATION IP BP USP Description Colorless crystals or white, Crystalline powder. White or almost white, crystalline powder or brilliant , colorless crystals. Colorless crystals or white, Crystalline powder Solubility – Freely Soluble in water , very soluble in boiling water and practically insoluble in ethanol (96 % )
Identification (A) Reaction
(B) Reaction
A : Reactions of Sulphates B : Reactions of Magnesium salts
A: It gives reaction of Sulphates. B :It gives reaction of magnesium
A: It gives reaction of Sulphates. B : It gives reaction of magnesium
pH of a 5% solution at 250C – – 5.0 – 9.2 Appearance of solution Dissolve 5 g in sufficient carbon dioxide free water to produced 50 ml (Solution A) is clear and colorless. Dissolve 5 g in sufficient carbon dioxide free water to produced 50 ml (Solution S) is clear and colorless. – Acidity or alkalinity NMT 0.2 ml of either 0.01 M hydrochloric acid or 0.01 M sodium hydroxide is required to change the color of solution. NMT.0.2 ml of 0.01 M hydrochloric or 0.01 M sodium hydroxide is required to change the color of indicator. – Arsenic NMT 2 ppm.
NMT 2 ppm – Heavy metals NMT 10 ppm.
– NMT 10 ppm Selenium – NMT 30 ppm Iron NMT 200 ppm. NMT 20 ppm NMT 20 ppm Chloride NMT. 300 ppm NMT. 300 ppm
NMT. 140 ppm Organic Volatile impurities – Meets the requirement Residual Solvents – Meets the requirement Loss on drying 48.0 to 52.0 %
48.0 % – 52.0 % NMT 2.0 % Loss on Ignition – – 40.0%-52.0% Assay (on dried basis) 99.0 % to 100.5 % 99.0 % – 100.5 % 99.0%- 100.5% Pigment Alpha Blue 15:0 ( CAS NO: 147-14-8 )
Technical SpecificationChemical NameCopper Phthalocyanine BlueColour index NamePigment Blue 15:0Colour index No.74160Molecular Weight575.5AppearanceBright Blue PowderpH6-8Sieve residue0.1% Max.Light FastnessFull Shade : 7-8Tint Shade : 7Heat Stability for 5 Minute140°CBulk Density0.45 g / vvMoisture Content0.5% Max.Pigment Blue 15:1
CAS NO: 147-14-8
TECHNICAL DATA OF ANALYSISProduct NamePigment Blue 15:1Chemical ClassPhthalocyanine BlueChemical FormulaC32H16N8CuMolecular Weight576C.I. Number74160Bulk density ( gm / cc )0.35 gm/ccPhysical AppearanceBlue Powderlspecific Gravity ( 2O° C )1.45 to 1.80pH of I0% Aq. Soln.7+=1Heat StabilityStable at 280° CMoisture Content<0.5%Sieve Residue ( on 325 BSS Mesh )<0.5%Solvent FastnessExcellentLight fastnessOut StandingChemical ResistanceExcellentPigment Blue 15:3
CAS NO: 147-14-8
Technical SpecificationChemical NameCopper Phthalocyanine BlueColour index NamePigment Blue 15:3Colour index No.74160Molecular Weight575.5AppearanceBright Blue PowderpH6-8Sieve residue0.1% Max.Light FastnessFull Shade : 7-8Tint Shade : 7Heat Stability for 5 Minute300°CBulk Density0.33 g / vvSolubility in Water0.25%Moisture Content0.5% Max.Solvent FastnessExcellentPigment Green 7
CAS NO: 1328-53-6
OTHER NAME: PHTHALOCYANINE GREEN G
TECHNICAL DATA OF ANALYSISProduct NamePigment Green-7Chemical ClassCopper Phthalocyanine GreenMolecular Weight1127C.I. Number74260Bulk density ( gm / cc )0.42 – 0.48Physical AppearanceGreen PowderSolubility in waterInsolubleOdorOdorlessSpecific Gravity ( 2O° C )1.5 to 1.6pH of I0% Aq. Soln.7+=1Heat Stability for 5 min.240° CMoisture Content<0.5%Sieve Residue ( on 325 BSS Mesh )<0.5%Solvent FastnessExcellentLight fastnessOut StandingChemical ResistanceExcellentPotassium Acetate
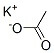
CAS NO: 127-08-2
MOLECULAR FORMULA :- C2H3KO2
MOLECULAR WEIGHT :- 98.14 g/mol
SPECIFICATION BP USP Description White or almost white, crystalline powder or colorless cryastals,deliquescent. White or almost white, crystalline powder or colorless cryastals,deliquescent. Solubility Very soluble in water, freely soluble in ethanol (96 %) – Identification A. Reaction
B. Reaction
A. Reaction of acetates.
B. Reaction of potassium
A. Reaction of acetates.
B. Reaction of potassium
Appearance of solution Clear & Colorless – pH 7.5-9.0 7.5-8.5 Heavy metals – NMT 20 ppm Limit of Sodium – NMT 0.03 % Reducing substances The solution remains pink. – Chloride NMT 200 ppm – Sulfates NMT 200 ppm – Aluminum NMT 1 ppm – Iron NMT 20 ppm – sodium NMT 0.5 % – Loss on drying NMT 3.0 % NMT 1.0 % Residual Solvents – Meets the requirements Assay 99.0 %-101.0 % 99.0%-100.5 % Potassium Carbonate Anhydrous
CAS NO: 584-08-7
MOLECULAR FORMULA :- K2CO3
MOLECULAR WEIGHT :- 138.21 g/mol
SPECIFICATION BP USP Description White or almost white granular Powder hygroscopic. White or almost white granular Powder hygroscopic. Solubility Freely Soluble in water, practically insoluble in Ethanol (96%) – Identification (A) Reaction
(B) Reaction
(C) Reaction
A : The solution is strongly alkaline B: Test A gives the reaction of Carbonates &Bicarbonates.
C: Test A gives reaction of Potassium.
A: Test A gives the reaction of Carbonates. B: Test A gives reaction of Potassium.
Insoluble substances – The solution is complete, clear & Colorless. Heavy metals – NMT 5 ppm Calcium NMT 100 ppm – Iron NMT 10 ppm – Chlorides NMT 100 ppm – Loss on Drying NMT 5.0 % NMT 0.5 % Sulphates NMT 100 ppm – Organic Volatile Impurities – Meets the requirement Residual Solvents – Meets the requirement Assay 99.0 % – 101 % (on dry basis) 99.5 % – 100.5 % Potassium Chloride
CAS NO: 7447-40-7
MOLECULAR FORMULA: KCl
MOLECULAR WEIGHT: 74.55
Alternate Names: Potassium Chloride is also known as KCl. Application: Potassium Chloride is a common laboratory reagent and calibration standard for measuring electrical conducivity. Purity: >99% Description: Potassium Chloride is commonly used as a laboratory reagent as a standard. Potassium Chloride may be used as a calibration standard ionic solution for measuring electrical conductivity because Potassium Chloride solutions, when prepared carefully, produce repeatable measurable properties. Potassium Chloride is also a good source of ionic chloride, which should precipitate insoluble chloride salts upon addition to a solution of an appropriate metal ion. Oral Potassium Chloride is commonly used to replenish vital potassium in the body, and has also been shown to lower blood pressure. Potassium Chloride is commonly used in fertilizer. Physical State : Solid Solubility : Soluble in water (340 mg/ml at 20° C), alcohol (slightly), and ethanol (1 g/250 mL). Insoluble in ether, and acetone. Storage : Store at room temperature Melting Point : 770° C (lit.) Boiling Point : 1500° C (lit.) Density : 1.98 g/cm3 at 25° C (lit.) Refractive Index : n20D 1.49 Ki Data : CA IV: Ki= 90 µM (human); CA I: Ki= 6 mM (human); CA IX: Ki= 33 mM (human); CA V: Ki= 156 mM (human); CA II: Ki>200 mM (human)
Showing 19–36 of 74 items